Metabolic Health Biomarkers: The Complete Testing Guide (+ My Tracker)
The blood tests your doctor isn't ordering, what low-risk ranges actually are, and the tool I built to track them over time and become an informed in my own health.
If you’re just joining, welcome!
Swiss Army Mum is a science-based wellness system for busy women. Four pillars - Body, Mind, Glow, Flow.
Not every tool, just the right ones.
Most metabolic health biomarkers aren’t tested until disease appears. By the time your doctor diagnoses prediabetes or metabolic syndrome, insulin resistance has been building for years. But there are blood tests that catch dysfunction early — fasting insulin, HOMA-IR, inflammatory markers — and most doctors don’t order them.
Over the past month, we’ve covered the Fuel pillar in depth: how to stabilize blood sugar, prevent insulin resistance, support your microbiome, and eat for long-term metabolic health. You learned how insulin resistance develops silently, years before fasting glucose looks abnormal.
So the obvious question: What should you actually test? How do you track metabolic health before it becomes a problem?
This post answers that. The biomarkers your doctor isn’t testing (but should be). What low-risk ranges are (versus “normal”). And a look at the Google Sheet I built to track them (it even has colors on it!)
Let’s close the Fuel pillar with a practical tool.
Why Standard Blood Tests Miss Metabolic Dysfunction
Your standard annual physical probably tests:
Fasting glucose
Total cholesterol, LDL, HDL
Triglycerides
Blood pressure
What’s missing:
Fasting insulin (the earliest indicator of insulin resistance)
HOMA-IR (insulin resistance calculation)
HbA1c (3-month glucose average)
Triglyceride:HDL ratio (insulin resistance proxy when fasting insulin isn’t available)
Inflammatory markers (hsCRP, uric acid)
Liver enzymes (AST, ALT, GGT — fatty liver indicators)
Why this matters:
Standard panels are designed to diagnose disease, not prevent it. The “normal” ranges are population averages - often based on populations with silent conditions. By the time a marker crosses into “abnormal,” you’ve often had dysfunction for years.
The SAM approach:
Test more comprehensively (annually at minimum, but not too often. lifestyle changes take a while to show in your bloodwork)
Track trends over time (not just single snapshots)
Aim for low-risk zones, not just “normal”
What 'Normal' Blood Sugar Really Means (And Why Low-Risk Zones Matter)
Before we dive into the biomarkers, let’s clarify what these terms mean:
“Normal” (Clinical Range): The range used by labs and doctors to diagnose disease. Often based on population averages that may include people with subclinical dysfunction. These are the thresholds that trigger a diagnosis or prescription. You want to stay away from this.
“Low-Risk Zone”: The range associated with lowest risk in population studies. This is where I focus — not just avoiding disease, but aiming for long-term metabolic health.
When I say “low-risk zone” or “optimization target,” I’m referencing research showing where risk is lowest — not an official medical guideline. This distinction matters.
Also important: some markers have ethnicity-specific thresholds (like waist circumference), and some vary by sex (like liver enzymes). The tables below reflect this where relevant.
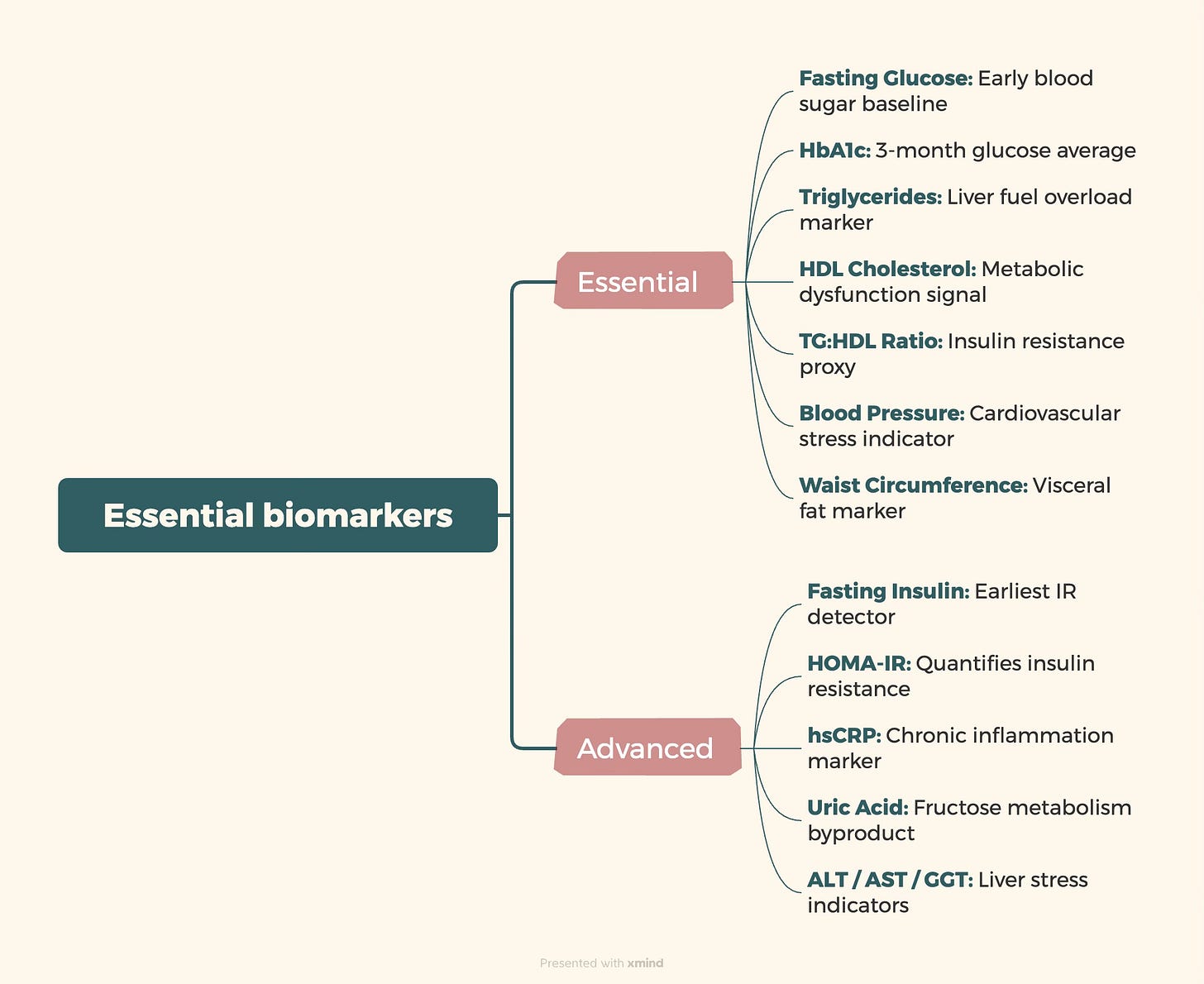
The Essential Biomarkers
One note on sources: the biomarker list in this post was partly inspired by Casey Means' Good Energy. I reviewed the book in my metabolic health reading list. I had real misgivings about its tone and some of its recommendations. But the biomarker framework she presents is genuinely useful and actionable, and it's grounded in solid underlying science. I cross-referenced every marker against primary research and clinical guidelines before including it here. That's the SAM approach: take what works, leave what doesn't.
I’m breaking this into two tiers:
Tier 1: Basic metabolic panel — essential, test annually
Tier 2: Advanced panel — highly recommended if accessible
Essential Metabolic Health Tests (Tier 1)
1. Fasting Glucose
Why it matters: Blood sugar after an overnight fast. It’s a lagging indicator — by the time it’s abnormal, you’ve had dysfunction for years. But even within the “normal” range, higher fasting glucose predicts diabetes risk.
Normal: <100 mg/dL (5.6 mmol/L)
Low-risk zone: 70-85 mg/dL (3.9-4.7 mmol/L)
Clinical data1: A study of over 46,000 people found that those with fasting glucose 95-99 mg/dL had significantly higher diabetes risk than those <85 mg/dL — despite both being “normal. The low end (70 mg/dL) is a standard clinical lower bound.
2. Hemoglobin A1c (HbA1c)
Why it matters:
Measures your 3-month average blood sugar by looking at the percentage of hemoglobin with sugar attached via glycation. More stable than fasting glucose and captures trends, not just snapshots.
Normal: <5.7%
Low-risk zone: 5.0-5.4%
Clinical data: The ARIC study (over 11,000 participants) used 5.0-5.4% as the reference category for heart failure risk, with risk increasing in categories above 5.5%2. Other large cohorts show similar patterns3. This isn’t an official target — it’s the category consistently associated with lowest risk in research.
3. Triglycerides
Why it matters: Triglycerides are fats carried in the bloodstream, largely packaged by the liver. When you eat more sugar and carbs than your liver can handle — especially fructose — the excess gets converted to fat and packaged as triglycerides. Elevated fasting triglycerides are therefore a well-established marker of metabolic dysfunction and are strongly associated with insulin resistance, metabolic syndrome, and non-alcoholic fatty liver disease.
Normal: <150 mg/dL (1.7 mmol/L)
Low-risk zone: Well below 100 mg/dL; <80 mg/dL often seen in very insulin-sensitive profiles
Clinical data : The clinical threshold (<150 mg/dL) is well-established. But research suggests that triglycerides in the “normal-high” range (100-150 mg/dL)4 may already be associated with inflammation and increased coronary risk.
4. HDL Cholesterol
Why it matters:
The “good” cholesterol that helps remove cholesterol from your bloodstream and carries it back to the liver for processing. Low HDL correlates with insulin resistance. The real signal is the pattern: low HDL + high triglycerides = atherogenic dyslipidemia, a hallmark of metabolic dysfunction.
Normal: Women >50 mg/dL (1.3 mmol/L), Men >40 mg/dL (1.0 mmol/L)
Desirable: ≥60 mg/dL (1.5 mmol/L)
How to interpret: Low HDL is clearly unfavorable. Higher HDL (≥60 mg/dL) has historically been considered protective, though recent studies show a U-shaped relationship in some populations — very high HDL isn’t always better5. Most useful when viewed alongside triglycerides (see next marker).
5. Triglyceride:HDL Ratio
Why it matters:
A simple calculation (triglycerides ÷ HDL, using mg/dL or mmol/L for both) that serves as an insulin resistance proxy when you don’t have access to fasting insulin testing. Studies show this ratio correlates well with underlying insulin resistance and can reveal dysfunction even when individual markers look “normal.”
Low-risk zone: <1.5 (lower is better)
Concerning: >3 strongly suggests insulin resistance
Clinical data: A ratio ≥3 identifies insulin-resistant individuals fairly well in many populations. Critical: if you’re using mmol/L instead of mg/dL, the numerical ratio will be different — you need to convert units6. A ratio >3 in mg/dL units corresponds to approximately >1.8 in mmol/L units.
6. Blood Pressure
Why it matters:
Measures the force of blood against your artery walls. High blood pressure is the most common preventable risk factor for cardiovascular disease, stroke, heart failure, kidney disease, and dementia. Chronic hyperinsulinemia raises blood pressure because insulin causes sodium retention.
Normal: <120/<80 mmHg
Clinical data: European guidelines define “optimal” as <120/<80 explicitly7 as optimal. A massive meta-analysis showed that cardiovascular risk increases continuously without an obvious threshold, at least down to 115/75 mmHg. Interpret with context: age, symptoms, pregnancy, medications.
7. Waist Circumference
Why it matters:
Proxy for abdominal fat, specifically visceral fat stored around your organs. Unlike subcutaneous fat (under the skin), visceral fat is metabolically active — it drives insulin resistance, inflammation, and metabolic dysfunction. Central adiposity predicts metabolic disease even when BMI is “normal.”
Normal (ATP III, often US): Women <88 cm (35 in), Men <102 cm (40 in)
Ethnicity-specific thresholds (International Diabetes Federation, IDF): European, Sub-Saharan African, Middle Eastern, Eastern Mediterranean: Women <80 cm (31.5 in), Men <94 cm (37 in)
How to interpret:
These aren’t “optimal” targets — they’re risk thresholds. The IDF cutoffs recognize that risk varies by ancestry and are widely used internationally8.
Advanced Insulin Resistance Tests (Tier 2)
8. Fasting Insulin
Why it matters:
THE earliest indicator of insulin resistance. Measures how much insulin your pancreas is pumping out after an overnight fast. Insulin rises years before glucose does — this is the marker that catches dysfunction earliest, often a decade before your doctor says “prediabetes.”
Normal: <25 mIU/L (varies by lab and population)
Low-risk zone: 2-5 mIU/L
Clinical data: There’s no universal consensus on cut-offs because methods vary, but patterns emerge: An NHANES analysis defined “hyperinsulinemia” as >10 µU/mL9; The 2-5 mIU/L range represents very insulin-sensitive profiles — it’s an optimization target, not a clinical guideline.
9. HOMA-IR (Homeostatic Model Assessment of Insulin Resistance)
Why it matters:
A calculation that combines fasting glucose and fasting insulin to quantify insulin resistance. More informative than either marker alone. Think of it as a snapshot of how hard your pancreas is working to keep glucose “normal” — high HOMA-IR means your body needs lots of insulin to manage blood sugar.
Formula: Formula: (Fasting Glucose in mg/dL × Fasting Insulin in mIU/L) ÷ 405 OR (Fasting Glucose in mmol/L × Fasting Insulin in mIU/L) ÷ 22.5
Low-risk zone: <1.0
Normal: <2.0
Insulin resistant: >2.5
Severely insulin resistant: >5.0
Clinical data: Cut-offs vary widely by population, age, sex, and ethnicity. The original HOMA paper notes day-to-day variability, reinforcing the value of tracking trends10. Bottom line: <2.0 is broadly consistent with “normal”; <1.0 represents very insulin-sensitive profiles.
10. High-Sensitivity CRP (hsCRP)
Why it matters:
Measures C-reactive protein, an inflammation marker made by your liver. Chronic low-grade inflammation drives metabolic dysfunction, cardiovascular disease, and accelerates aging (”inflammaging”). High hsCRP indicates your body is in a persistent inflammatory state, which damages tissues and worsens insulin resistance over time.
Risk categories (AHA/CDC):
Low risk: <1.0 mg/L
Average risk: 1.0-3.0 mg/L
High risk: >3.0 mg/L , higher suggests acute inflammation
11. Uric Acid
Why it matters:
A metabolic byproduct of fructose metabolism and purine breakdown (from meat and alcohol). Elevated uric acid is associated with metabolic dysfunction, gout, hypertension, and cardiovascular disease. It’s both a marker of metabolic stress and potentially a contributor to it - high levels can damage blood vessels and worsen insulin resistance.
Normal: Women 1.5-6.0 mg/dL, Men 2.5-7.0 mg/dL
Low-risk zone (optimization target): Women 2.0-4.0 mg/dL, Men 3.0-5.5 mg/dL
How to interpret:
Serum becomes saturated in urate around 6.8 mg/dL, favoring crystallization (gout)11. But risk increases gradually even within “normal” ranges. A large cohort found that hypertension risk increases with uric acid starting as low as 4 mg/dL in some subgroups12. Caveat: Causality is debated — uric acid may be a marker rather than a cause.
12. Liver Enzymes: AST, ALT, GGT
Why they matter:
Proteins released when liver cells are damaged or inflamed. Elevated enzymes suggest fatty liver (NAFLD), the most common chronic liver disease and a hallmark of metabolic dysfunction. They catch liver stress before clinical disease appears.
Normal (typical lab ranges):
ALT: 7-55 U/L
AST: 8-48 U/L
GGT: 6-61 U/L
Low-risk zone (proposed “healthy normal”):
ALT: Women ~19-25 U/L, Men ~29-33 U/L
AST: Similar to ALT
GGT: Women <20 U/L, Men <25 U/L
How to interpret:
Standard lab upper limits have been criticized because they include people with silent liver disease. A landmark study proposed lower upper limits for ALT (~30 men, ~19 women) in truly healthy populations13.
How to Read Your Metabolic Health Results
Metabolic dysfunction doesn’t happen overnight. It develops slowly, over years or even decades. The point of tracking these biomarkers isn’t to obsess over every tenth of a point or panic when one number isn’t perfect.
The goal is awareness and trends.
One abnormal result doesn’t mean disaster. Biomarkers fluctuate based on hydration, stress, sleep quality, recent meals (even when fasting), menstrual cycle, and more. Single data points can be noisy.
Trends matter more than snapshots. Test annually. Are your markers moving in the right direction or stable? That’s what counts.
Lab ranges vary. Different laboratories use different methods and reference populations. What’s “normal” at one lab may differ slightly at another - especially for insulin and liver enzymes.
Individual variation is real. Age, sex, ethnicity, genetics, and activity level all affect what’s “normal” for you. The ranges below are population-based guidelines, not hard rules.
Use this as a tool, not a source of anxiety. The tracker makes the invisible visible so you can see whether the Fuel principles (and Sleep, Exercise) are working. It’s feedback, not judgment.
If results are concerning or you’re unsure how to interpret them, work with a healthcare professional. This is educational information, not medical advice.
Why Tracking Your Own Biomarkers Empowers Better Health Decisions
Here’s what changed for me when I started tracking these markers: I stopped being a passive patient.
For years, I’d go to my annual physical, get a printout with numbers I didn’t fully understand, and my doctor would say “everything looks normal” or “your cholesterol is a bit high, let’s recheck in six months.” I’d leave with vague anxiety and no clear action plan.
Now? I understand what the numbers mean. I know which markers predict future problems years before diagnosis. I can ask specific questions.
This isn’t about replacing my doctor. It’s about being an informed partner in my own health. That’s empowering.
My doctor is the expert. She has medical training, clinical experience, and context I don’t have. She can interpret results in light of my full health history, prescribe medication when needed, and catch things I’d miss.
But I am the expert on my body and my daily choices. I’m the one implementing the Fuel principles. I’m the one tracking sleep, managing stress, exercising consistently. I’m the one who sees patterns the lab can’t capture.
When I bring data, not just one test result, but trends over time, my doctor and I can have deeper, more productive conversations. We can make decisions together based on evidence, not guesswork.
The goal isn’t perfection. It’s agency.
The Metabolic Health Tracker (Google Sheets Template)
That's why I built this tracker - not to replace medical expertise, but to make myself a better partner in my own health. I wanted to see patterns, not just single snapshots. I wanted to know if the Fuel, Sleep and Exercise principles were actually working. I wanted a visual reference I could return to annually.
What’s included in the tracker:
All biomarkers listed above
Date tracking (annual testing with trend visualization)
Automatic color-coding (green = low-risk zone, yellow = normal but not optimal, red = out of range)
HOMA-IR auto-calculation (input fasting glucose + insulin → calculates HOMA-IR)
Triglyceride:HDL ratio auto-calculation
Two sheets for SI and US units
How it works:
Input test date + values
Sheet auto-calculates HOMA-IR and TG:HDL ratio
Cells color-code based on low-risk/normal/out-of-range
Note: This tracker will be available to paid subscribers when I launch the Swiss Army Mum Toolkit later this year. For now, I’m showing you what it looks like and why it works. But you can also make one yourself! This is the made-for-you option.
My results
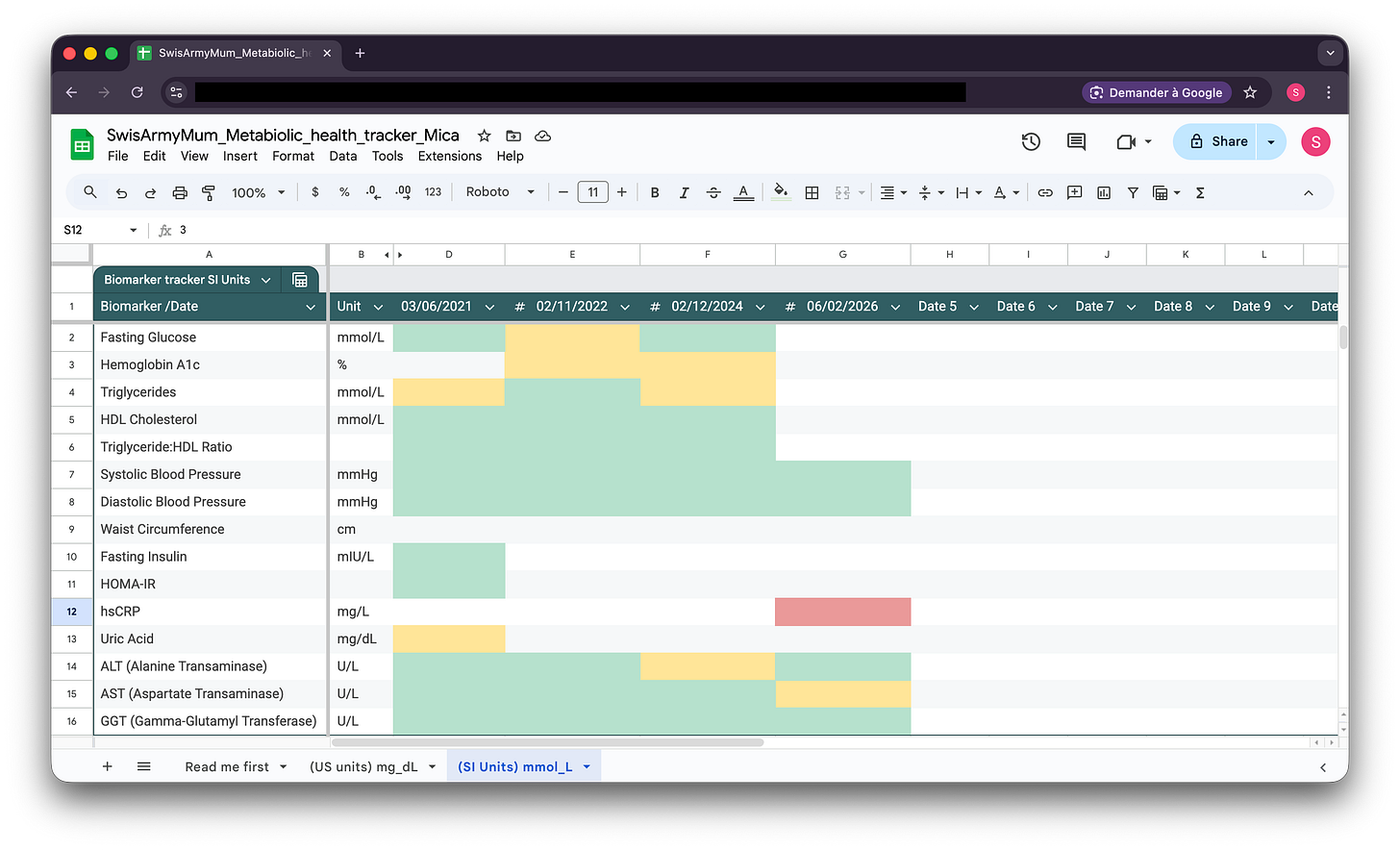
When I look at this tracker, I don’t see numbers. I see patterns.
Green dominates. That means my metabolic system isn’t quietly deteriorating. The markers that predict dysfunction years in advance - fasting insulin, HOMA-IR, triglyceride:HDL ratio - aren’t creeping into yellow or red. They’re stable in the low-risk zone.
Blood sugar control is holding. Fasting glucose and HbA1c haven’t drifted upward over time. No slow escalation. No quiet progression toward prediabetes. The system that regulates glucose isn’t under chronic stress.
Lipids are coordinated. Triglycerides and HDL are behaving like they belong in the same metabolic system - which they do. When those two move together in the right direction, it usually reflects how efficiently your liver handles fuel over the long term. Not one meal. Not one week. The trend.
The insulin markers matter most to me. Fasting glucose can stay “normal” for years while insulin quietly climbs in the background, compensating. I only have one measurement of fasting insulin. I’ll make sure to get another one at my next physical.
Blood pressure is stable. No progressive tightening. No upward creep. A cardiovascular system that doesn’t look chronically irritated.
There’s one red cell. Inflammation spiked once. And I have context for that - I was recovering from an acute illness when I tested. Inflammatory markers are supposed to respond to acute stress. What would concern me is persistent elevation across multiple time points without explanation, which is not what I see. I wish I had tested hRCP on a different occasion, will make sure to add that one too next time.
Liver enzymes fluctuate slightly between tests. That’s normal biology. Training intensity, illness, stress, even a disrupted week can move them. What matters is trajectory - are they climbing progressively over time? No. Just normal variation within a stable range.
That’s the theme across this tracker: stability, not perfection.
No silent deterioration. No quiet metabolic unraveling.
I still have pizza nights. I still have dessert. I still live like a human. But I don’t graze all day. I prioritize protein and fiber. I lift weights. I walk after meals. I sleep like my life depends on it. And when I zoom out across years of data, the patterns reflect that.
The Fuel principles aren’t about perfect labs. They’re about a metabolism that doesn’t quietly break down over time.
What the Tracker Can’t Tell You (Limitations)
This Isn’t a Substitute for Medical Care
If results are abnormal, work with a doctor.
Don’t diagnose yourself or self-treat based on spreadsheet colors.
Some conditions require medication (e.g., very high BP, diabetes) — lifestyle isn’t always enough.
Individual Variation Exists
Low-risk zones are population-based guidelines, not hard rules.
Genetics, ancestry, age, sex, activity level all affect what’s “normal” for you.
Use the tracker as a guide, not gospel.
Single Tests Can Be Noisy
Hydration, stress, sleep quality, recent meals (even if fasting), menstrual cycle — all affect results.
One abnormal result doesn’t mean panic.
Trends matter more than single data points.
What’s Next
We’ve now covered Sleep and Fuel in depth — 2 out of 3 Body sub-pillars.
Next: Exercise: muscle as the longevity organ, the 80/20 workout routine, and why strength training matters more than you think.
See you February 25.
Subscribe (free) so you don’t miss Exercise!
Thank You
Thank you for reading, sharing, and supporting this work. Whether you’ve been here since the beginning or just found Swiss Army Mum, I’m glad you’re here.
Building sustainable health without overwhelm takes a village. If something resonated with you, I’d be grateful if you forwarded this to someone who might benefit or hit the ♥️ or ↻ Restack button. It helps more people discover this space.
See you next week.
— Mica
Your Turn
Have you had comprehensive metabolic testing done? What surprised you?
Which biomarker are you most interested in tracking?
If you’ve been following the Fuel principles, have you retested to see if they’re working?
Comment below — I read every one.
Medical note: This is educational, not personal medical advice. Your biology, history, and context matter. Work with a qualified healthcare professional.
Nichols, Gregory A et al. “Normal fasting plasma glucose and risk of type 2 diabetes diagnosis.” The American journal of medicine vol. 121,6 (2008): 519-24. https://doi.org/10.1016/j.amjmed.2008.02.026.
Matsushita, Kunihiro et al. “The association of hemoglobin a1c with incident heart failure among people without diabetes: the atherosclerosis risk in communities study.” Diabetes vol. 59,8 (2010): 2020-6. https://doi.org/10.2337/db10-0165.
Selvin, Elizabeth et al. “Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults.” The New England journal of medicine vol. 362,9 (2010): 800-11. https://doi.org/10.1056/NEJMoa0908359.
Miller, Michael et al. “Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association.” Circulation vol. 123,20 (2011): 2292-333. https://doi.org/10.1161/CIR.0b013e3182160726.
Liu, Chang et al. “Association Between High-Density Lipoprotein Cholesterol Levels and Adverse Cardiovascular Outcomes in High-risk Populations.” JAMA cardiology vol. 7,7 (2022): 672-680. https://doi.org/10.1001/jamacardio.2022.0912.
Brehm, Attila et al. “Relationship between serum lipoprotein ratios and insulin resistance in obesity.” Clinical chemistry vol. 50,12 (2004): 2316-22. https://doi.org/10.1373/clinchem.2004.037556.
Williams, Bryan et al. “2018 ESC/ESH Guidelines for the management of arterial hypertension.” European heart journal vol. 39,33 (2018): 3021-3104. https://10.1093/eurheartj/ehy339.
Alberti, K G M M et al. “Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity.” Circulationvol. 120,16 (2009): 1640-5. https://doi.org/10.1161/CIRCULATIONAHA.109.192644
Wu, Chuyue et al. “Trends in Hyperinsulinemia and Insulin Resistance Among Nondiabetic US Adults, NHANES, 1999-2018.” Journal of clinical medicinevol. 14,9 3215. 6 May. 2025, https://doi.org/10.3390/jcm14093215
Matthews, D R et al. “Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man.” Diabetologia vol. 28,7 (1985): 412-9. https://doi.org/10.1007/BF00280883.
Kang, Duk-Hee, and Sung-Kyu Ha. “Uric Acid Puzzle: Dual Role as Anti-oxidantand Pro-oxidant.” Electrolyte & blood pressure : E & BP vol. 12,1 (2014): 1-6. https://doi.org/10.5049/EBP.2014.12.1.1.
Grayson, Peter C et al. “Hyperuricemia and incident hypertension: a systematic review and meta-analysis.” Arthritis care & research vol. 63,1 (2011): 102-10. https://doi.org/10.1002/acr.20344.
Prati, Daniele et al. “Updated definitions of healthy ranges for serum alanine aminotransferase levels.” Annals of internal medicine vol. 137,1 (2002): 1-10. https://doi.org/10.7326/0003-4819-137-1-200207020-00006.