The Insulin Resistance Story (And My Glucose Monitor Data)
How insulin resistance actually develops, why your body responds differently than mine, and what two weeks of continuous glucose data revealed about my metabolism.
Last week, I published the Fuel blueprint - the 80/20 of eating for metabolic health: stabilizing blood sugar, preventing insulin resistance, prioritizing whole foods, and letting insulin do what it’s designed to do.
This post is not a continuation of that blueprint.
This is a Zoom post: the story underneath it.
No protocols. No hacks. No food rules.
Just mechanism, context, and a better way to think about metabolism.
You Can Have Normal Glucose and Still Be Metabolically Broken
For decades, metabolic health has been treated as something you can assess with a single number.
If fasting glucose is “normal,” you’re fine.
If it isn’t, you’re not.
That framing is comforting - and deeply misleading.
Fasting glucose is measured at the most forgiving moment possible: after an overnight fast, when insulin has already had hours to quietly clean up the system. It tells you almost nothing about how metabolism behaves the rest of the day — after meals, under stress, with imperfect sleep, in real life.
You can have completely normal fasting glucose and still be metabolically dysfunctional.
Fasting glucose is a lagging indicator. By the time it becomes abnormal (crosses 100 mg/dL), you’ve likely had insulin resistance for a decade. The damage starts much earlier - when your body is pumping out excess insulin to keep glucose “normal.”
And your doctor usually won’t tell you to do anything until a guideline value prompts a prescription drug - for life.
This post is about that story.
How insulin resistance actually develops.
Why fructose plays a disproportionate role.
Why identical foods produce radically different responses.
And what my CGM data taught me about my own metabolism.
New here? Start here:
Swiss Army Mum is a practical guide to long-term health for busy women, built on four pillars: Body, Mind, Glow, and Flow.
Not every tool. Just the right ones.
The Insulin Mechanism: How Metabolic Dysfunction Actually Develops
What Insulin Does
Insulin is a storage hormone. When you eat, your blood glucose rises. Your pancreas releases insulin to shuttle that glucose into cells (muscle, liver, fat) where it can be used for energy or stored for later.
In a healthy metabolism, this works beautifully. Glucose goes up, insulin goes up, glucose gets cleared from the bloodstream, insulin drops back down.
Clean, efficient, no problems.
But when you eat foods that spike glucose repeatedly - refined carbohydrates, added sugars, especially liquid sugar - your cells get flooded with fuel over and over.
Eventually, they stop responding to insulin’s signals.
This is insulin resistance.
The Progression: From Healthy to Dysfunctional
Here’s how it unfolds, often over decades:
Stage 1: Normal metabolism
You eat, glucose rises, insulin rises, glucose clears, insulin drops
Cells respond properly to insulin signals
Everything works as designed (being young has its perks!)
Stage 2: Early insulin resistance (silent phase)
You eat, glucose rises, but cells don’t respond as well to insulin
Pancreas compensates by producing MORE insulin to get the same glucose-clearing effect
Fasting glucose still looks normal (because the extra insulin is doing its job)
But you now have hyperinsulinemia - chronically elevated insulin levels
This stage can last years. You feel fine. Your doctor says your glucose is normal. But insulin is quietly doing damage: promoting fat storage, raising your body-weight set-point, triggering inflammation.
Stage 3: Advanced insulin resistance
Cells are significantly insulin resistant
Pancreas pumps out even more insulin, but it’s not enough anymore
Post-meal glucose stays elevated longer (you’d see this on a CGM, but not on a fasting test)
Stage 4: Prediabetes/Type 2 Diabetes
Pancreas can’t keep up anymore
Fasting glucose finally crosses 100 mg/dL (prediabetes) or 130 mg/dL (diabetes)
Now it shows up on standard tests - but the damage has been accumulating for decades
The critical insight from Jason Fung’s The Obesity Code: By the time your fasting glucose is abnormal, you’re already deep into metabolic dysfunction. Insulin resistance starts long before glucose becomes a problem.
Why Insulin Matters More Than Glucose
Glucose is the symptom. Insulin is the cause.
When insulin is chronically elevated:
Fat storage increases. Insulin signals your body to store energy as fat and blocks fat breakdown (lipolysis). You literally cannot access stored fat for energy when insulin is high.
Your set-point rises. Your body defends a higher weight because insulin has recalibrated what it considers “normal.”
Cells starve despite abundance. Insulin-resistant cells can’t take in glucose properly, so they’re energy-deprived even though your bloodstream is full of fuel. Meanwhile, excess glucose gets shunted into fat storage.
Inflammation increases. Hyperinsulinemia triggers inflammatory pathways, worsening the feedback loop.
This is why calorie restriction often fails for weight loss. If insulin is high, your body stays in fat-storage mode no matter how little you eat. You can’t out-starve a hormonal problem.
Casey Means’ perspective from Good Energy: Chronic hyperinsulinemia also damages mitochondria - your cellular powerhouses. Damaged mitochondria produce less ATP (energy) and more free radicals (oxidative stress), which further drives inflammation and insulin resistance. It’s a vicious cycle: insulin resistance → mitochondrial damage → more oxidative stress → worsening insulin resistance.
The Two-Compartment Model
Think of your body as having two compartments:
Bloodstream (where glucose circulates)
Cells (where glucose should go for energy)
In a healthy metabolism, glucose moves easily from bloodstream to cells. Insulin opens the door.
In insulin resistance, the door is stuck. Glucose piles up in the bloodstream (eventually leading to high blood sugar), but cells are starving. Your body’s solution? Store the excess glucose as fat.
This is why people with insulin resistance often feel tired despite eating plenty. Their cells aren’t getting fuel efficiently.
Swiss Army Mum angle
This insulin-driven fat storage mechanism is why Principle 2 from the Fuel blueprint (cut added sugars and refined grains) matters so much. Refined carbs and sugars spike glucose repeatedly, forcing your pancreas to pump out insulin over and over. Eventually, you develop insulin resistance. And why Principle 5 (space your meals and fast overnight) is critical - constant grazing keeps insulin chronically elevated, never giving your body a chance to drop into fat-burning mode.
Fung's work taught me that metabolic health isn't about willpower or calorie restriction. It's about managing the hormonal signals that control fat storage.
Fructose: The Hidden Villain
If insulin resistance is the fire, fructose is the gasoline.
How Fructose Differs from Glucose
Glucose and fructose are both sugars, but they’re metabolized very differently.
Glucose:
Enters bloodstream → triggers insulin release → shuttled into cells (muscle, liver, fat) for energy or storage
Every cell in your body can use glucose
Regulated by insulin
Fructose:
Bypasses normal glucose regulation entirely
Goes straight to the liver (the only organ that can metabolize it in significant amounts)
Rapidly converted to liver fat through a process called de novo lipogenesis1 (literally “making new fat”)
Does NOT trigger insulin release (which sounds good but isn’t - it means fructose sneaks past your body’s regulatory mechanisms)
Why Liquid Sugar Is Worse
When you eat an apple, you get fructose + fiber. The fiber slows absorption, feeds your gut bacteria, and moderates the glucose/insulin response. Your liver can handle the fructose load from one apple.
When you drink a glass of orange juice, you get the fructose of 4-5 oranges with ZERO fiber. Your liver gets slammed with fructose all at once. It converts the excess to fat and stores it directly in the liver - hello, non-alcoholic fatty liver disease (NAFLD).
Your liver doesn’t distinguish between fresh-pressed juice and a can of Coke. Both deliver a fructose bomb without fiber.
From Jason Fung’s The Obesity Code: Liquid sugar - soda, juice, sweetened lattes, energy drinks - is the single fastest route to insulin resistance and fatty liver. The combination of glucose (spikes insulin) and fructose (creates liver fat) is metabolically catastrophic.
The “Fruit Is Fine, Juice Is Not” Rule
Whole fruit: fiber buffer + micronutrients + polyphenols = moderate, manageable fructose load
Fruit juice: fructose bomb + no fiber = metabolic disaster
Dried fruit: concentrated fructose + no water content = easy to overeat, blood sugar spike
If you’re going to eat fruit (and you should!), eat it whole. Skip the juice.
Swiss Army Mum angle
This is why liquid sugar - soda, juice, sweetened lattes - is explicitly called out in Principle 2 of the Fuel blueprint. It's not just that they're "empty calories." It's that fructose bypasses your body's regulatory mechanisms and creates liver fat directly.
And why Nassim Taleb's rule (consume drinks that are at least 1,000 years old) isn't just clever - it's metabolically sound. Water, tea, coffee, wine, kombucha - these drinks don't assault your liver with fructose.
Most everything invented in the last century does.
Why Your Body Responds Differently Than Mine
Now here’s where it gets interesting.
I just explained the insulin mechanism (from Fung) and the fructose problem (from Fung and Means). These are universal - they apply to everyone. But HOW MUCH any given food spikes YOUR glucose depends on factors unique to you.
This is where Eran Segal and Eran Elinav’s research for The Personalized Diet becomes critical. Their research involved putting continuous glucose monitors on 800+ people and tracking their responses to identical foods.
The results? Wildly different.
One person’s glucose spiked from bananas but stayed flat after ice cream. Another person showed the exact opposite pattern. Same foods, opposite responses.
What Determines Your Unique Response?
1. Genetics
Some people have genetic variants that affect insulin sensitivity, glucose metabolism, and fat storage
You can’t change your genetics, but you can work with them
2. Gut microbiome composition
Beneficial bacteria ferment fiber into short-chain fatty acids (SCFAs) that improve insulin sensitivity
Harmful bacteria produce inflammatory metabolites that worsen insulin resistance
Your microbiome composition determines how you process specific foods
3. Sleep quality
Poor sleep increases insulin resistance acutely (even one bad night impairs glucose tolerance)
Chronic sleep deprivation worsens it long-term
4. Stress levels
Cortisol (stress hormone) raises blood glucose and worsens insulin sensitivity
Chronic stress = chronically elevated cortisol = metabolic dysfunction
5. Activity level and muscle mass
Muscle is a glucose sink
More muscle = better glucose disposal = lower insulin demand
6. Meal timing and circadian rhythm
Insulin sensitivity is highest in the morning, lowest in the evening (as melatonin rises)
Eating the same meal at 8 AM vs. 8 PM produces different glucose responses
The Limitation of Universal Dietary Advice
This is why rigid dietary dogma fails. “Eat this, not that” advice assumes everyone responds the same way. We don’t.
The fundamentals still apply universally - whole foods, stable glucose, adequate protein, healthy fats, plenty of fiber. But the details (rice vs. potatoes, oatmeal vs. eggs, intermittent fasting vs. regular meals) are individual.
The only way to know how YOUR body responds is to test. Either with a CGM (if accessible) or with careful symptom tracking (energy levels, cravings, mood, how you feel 2-3 hours post-meal).
Which brings me to my own experiment.
My CGM Experiment: What Two Weeks of Data Taught Me
I wore a continuous glucose monitor for two weeks as part of the FoodAndYou clinical trial at EPFL. The goal wasn’t optimization - I did this before reading deeply about glucose hacks - it was observation.
Inspired by the work of Fung, Segal and Elinav, I wanted to see how my body actually responded to the foods I eat regularly.
It was actually super fun to look at this data.
Here are the four key lessons.
Lesson 1: My Breakfasts Are On Point
What I eat most mornings:
Skyr/Greek yoghurt parfait with fruits (kiwi, passion fruit, frozen berries), nuts/nut butter, chia seeds/flax seed, psyllium husks, cacao nibs.
Chia pudding with a coulis of berries and nut butter.
Glucose response: Flat. Look at this graph where you see the days I ate my breakfasts (green) vs. the days I had to eat the “standard breakfasts” for the study (red - white bread alone, with butter or a standard glucose drink - yuk):
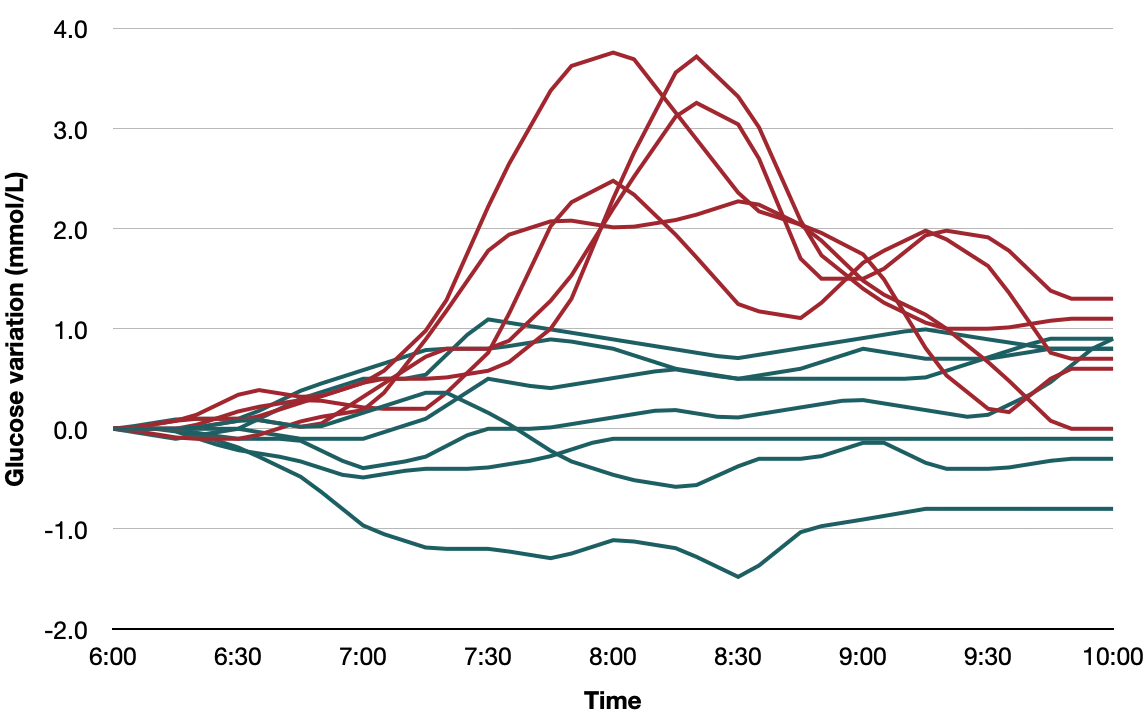
Why this works: Protein + fat + fiber - a perfect combo for stabilizing blood sugar and feeling full. I can go 3-4 hours easily after these breakfasts.
Lesson 2: Sweet Potatoes Spike Me (Sadly)
What happened: I love sweet potatoes. I thought they were a “healthy carb” and ate them regularly - roasted, mashed, in bowls.
Glucose response: Spike. Every. Single. Time.
Even when I added protein and fat (grilled chicken, olive oil), sweet potatoes still spiked me significantly. Not as high as eating them alone, but still well above my baseline.
Why this surprised me: Sweet potatoes have a lower glycemic index than white potatoes (in theory). But clearly, my body doesn’t handle them well.
What I changed: I didn’t eliminate sweet potatoes entirely - I still enjoy them occasionally. But I stopped treating them as a staple. I swapped them for other complex carbs (mostly legumes) that my body handles better.
Takeaway: “Healthy” is individual. Sweet potatoes might be fine for you. They’re not optimal for me. This is why personalized data matters.
Lesson 3: Movie Night Is a Metabolic War Zone (But I Still Do It)
Friday night ritual: Pizza + ice cream + a movie with my family.
Non-negotiable.
This is quality time, connection, joy. I’m not giving it up for perfect glucose curves.
Glucose response during the trial: Unmitigated disaster. Several huge peaks. Stayed elevated for hours. Glucose rollercoaster overnight (While I was sleeping!). Take a look at this graph showing several “normal nights” where glucose levels went back to baseline and stayed low overnight (green lines) vs. a typical movie night while wearing the CGM (red).
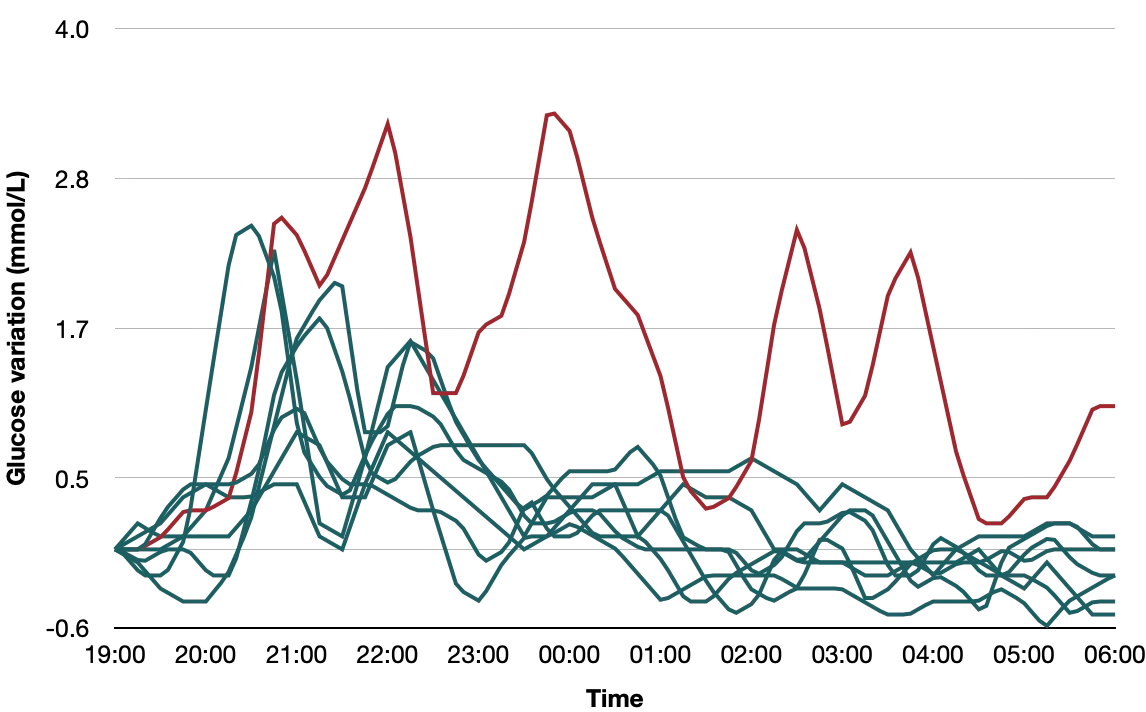
What I changed (after the trial):
Swapped store-bought ice cream for homemade 'nice cream’ - frozen berries blended with Greek yogurt. Same creamy, cold, sweet experience. Way less sugar, way more protein.
Walk on the treadmill for a while after eating. Not a workout—just a casual walk while we’re watching the movie. It helps me sleep better afterwards!
Takeaway: You don’t have to eliminate all foods you love or sacrifice family rituals for metabolic health. You should just be mindful about them and try to minimize the impact. As long as you’re not doing this every day, you’re OK!
Lesson 4: Biphasic Glucose Peaks
What I noticed: instead of a single rise and fall after certain meals, my glucose curve began to show two distinct peaks: a first rise, a drop, then a second, smaller rise before returning to baseline.
And sometimes three, as seen on the graph below!
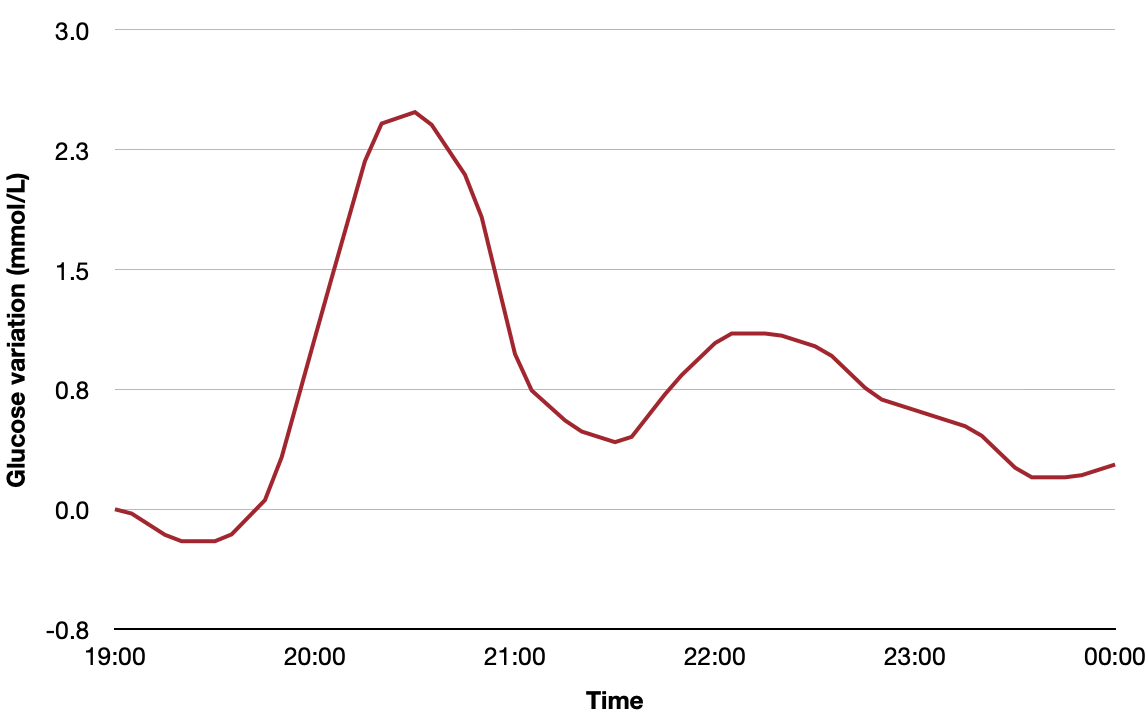
At first glance, this looks like poor control - delayed digestion, unstable glucose, something “off”.
Actually, this pattern is frequently associated with better insulin sensitivity, not worse.
Multiple studies using oral glucose tolerance tests show that individuals with monophasic curves tend to have impaired early insulin secretion, higher insulin resistance, worse long-term metabolic outcomes, whereas biphasic curves are associated with more efficient glucose handling and healthier beta-cell function23.
Takeaway: Metabolism is complex and individual. Standard dietary advice can’t account for this level of nuance. If something doesn’t make sense, investigate. Test. Stay curious.
Should You Try a CGM?
Honestly? Probably not necessary for most people.
CGMs are expensive and the data can be overwhelming if you don’t know how to interpret it.
A CGM is worth considering if:
You’re curious about your personal responses to specific foods
You’ve hit a plateau and want to troubleshoot what’s sabotaging you
You have access through a clinical trial (like I did) or your doctor prescribes one
You’re genuinely interested in self-experimentation and have the budget
Low-tech alternative: Track how you feel 2-3 hours after meals. Do you crash? Crave sugar? Feel foggy? That’s your body telling you the meal spiked your glucose. You don’t need a device to know when something’s off.
The 10 Glucose Hacks (From Jessie Inchauspé’s Glucose Revolution)
Now that you understand the insulin mechanism and why individual responses vary, let’s talk strategy.
Jessie Inchauspé’s Glucose Revolution distills years of glucose research into ten practical, evidence-based hacks. These aren’t theoretical - they’re actionable tactics you can implement today.
I didn’t optimize for these during my initial CGM trial (I read the book afterward), sadly. Who knows - I might re-do the experiment just to test them out systematically!
Here are all ten, with brief explanations:
1. Eat foods in the right order
Vegetables first, then protein and fat, then starches and sugars last.
Why it works: Fiber from vegetables creates a physical barrier in your gut that slows glucose absorption from the rest of your meal. Same foods, different order, dramatically different glucose response.
2. Add a veggie starter to all your meals
Even a small salad, some raw carrots, or steamed broccoli before your main course makes a difference.
3. Flatten your breakfast curve
Savory beats sweet every single time.
A sweet breakfast (toast, cereal, fruit smoothie) sets you up for glucose rollercoaster all day. A savory breakfast (eggs, vegetables, protein) stabilizes glucose and reduces cravings for hours.
4. Have any type of sugar you like - they’re all the same
Your body doesn’t distinguish between honey, agave, coconut sugar, white sugar, or maple syrup. They all spike glucose and insulin.
Don’t fall for “natural” sugar marketing. Sugar is sugar.
5. Pick dessert over a sweet snack
If you’re going to eat something sweet, eat it immediately after a meal rather than as a standalone snack.
Why: The fiber, protein, and fat from your meal slow the absorption of sugar. Eating dessert alone spikes you harder.
This doesn’t mean eat dessert every day - but if you’re going to have it, timing matters.
6. Reach for vinegar before you eat
1-2 tablespoons of vinegar (diluted in water or as salad dressing) before a meal blunts the glucose spike by ~20-30%.
Acetic acid slows gastric emptying and improves insulin sensitivity. Simple, cheap, evidence-backed.
7. After you eat, move
A 10-minute walk within 30 minutes of eating drops post-meal glucose measurably.
Why: Muscles absorb glucose directly from the bloodstream without needing insulin.
8. If you have to snack, go savory
Nuts, cheese, veggies with hummus, hard-boiled eggs - all better choices than fruit, crackers, granola bars, or anything sweet.
Better yet: don’t snack. Space meals 3-4 hours apart and let your insulin drop between meals.
9. Put some clothes on your carbs
Never eat naked carbs. Always add protein, fat, or both.
Toast → toast + avocado + egg Banana → banana + almond butter Rice → rice + salmon + olive oil
10. Stop counting calories
Calories matter for energy balance, but focusing on glucose stability is more important for metabolic health.
A 300-calorie muffin spikes glucose and insulin. A 300-calorie meal of eggs, avocado, and vegetables doesn’t.
Same calories, completely different metabolic outcome.
Which Hacks Made the 80/20?
When I was building the Fuel blueprint, I synthesized Inchauspé’s ten hacks alongside Fung’s insulin framework and Gottfried’s hormonal protocols. Here’s how they map:
Hacks 1 & 2 (food order, veggie starters) → Principle 1 & 4 (whole foods and eat plenty of fiber). Fiber first isn’t just about satiety - it’s about creating a physical barrier that slows glucose absorption.
Hack 10 (never eat naked carbs) → Principle 3 (eat enough protein, add natural fats). Protein and fat blunt the glucose spike, keeping insulin lower.
Hack 7 & 8 (vinegar, move after eating) → Advanced optimization. Solid evidence, easy to implement, but not essential if you’re doing Principles 1-5 consistently.
Hacks 3, 4, 5, 6, 9 → Principle 2. Understanding WHY savory breakfast beats sweet, or WHY dessert-after-meal beats standalone snack, reinforces the principles without adding complexity.
The bottom line: The Fuel blueprint includes the most impactful interventions from Inchauspé’s work (and Fung’s, and Gottfried’s) into five core principles. Master those first.
Add the tweaks as you’re ready.
When Keto Makes Sense (And When It Doesn’t)
Let’s talk about the Gottfried Protocol from Women, Food, and Hormones.
Dr. Sara Gottfried designed a four-week ketogenic reset specifically for perimenopausal and menopausal women whose bodies stopped responding to traditional approaches.
The structure:
Week 1 (Prep): Detox with cruciferous vegetables, high fiber, gentle carb reduction
Weeks 2-4 (Implementation): Strict ketogenic eating (net carbs <25g/day, ketogenic ratio 2:1 fat-to-protein+carbs)
Transition: Gradual return to Mediterranean-style anti-inflammatory diet
Done 2x per year.
Who It’s For
This is NOT a first step. It’s for women who:
Are in perimenopause or menopause
Have tried the 80/20 consistently for months without results
Are experiencing stubborn metabolic resistance (can’t lose weight, constant fatigue, brain fog despite “doing everything right”)
Have medical supervision or guidance
Why Short-Term Keto Can Reset Insulin Sensitivity
Declining estrogen and progesterone make women more vulnerable to insulin resistance. Sometimes standard approaches aren’t enough.
A brief ketogenic phase forces your body to switch from glucose-burning to fat-burning (ketosis). This gives your insulin pathways a break, reduces liver fat, and can improve insulin sensitivity when you transition back to moderate carbs.
Key: It’s a RESET, not a lifestyle. Chronic keto has risks (nutrient deficiencies, hormonal disruption, loss of metabolic flexibility).
Swiss Army Mum angle
How this fits the Fuel blueprint: The Gottfried Protocol isn't part of the 80/20 because it's not for everyone. It's advanced optimization for a specific population - perimenopausal and menopausal women with stubborn metabolic resistance despite doing the fundamentals consistently. If Principles 1-5 are working, you don't need this. But if declining hormones have made standard approaches ineffective, Gottfried's short-term keto reset offers a science-backed intervention. I included it in "The Next Level" section of the blueprint for exactly this reason - it's a tool, not a foundation.
What’s Next
Next week: Genius Foods Deep Dive—the 10 foods that protect your brain while fixing your metabolism. Why wild salmon matters, what broccoli does to activate your body’s master antioxidant switch, and how to prioritize them without overthinking.
After that: Kombucha and gut health - how fermented foods support the metabolic allies we talked about in the Fuel blueprint.
Subscribe (free) so you don’t miss it.
Thank You
Thank you for reading, sharing, and supporting this work. Whether you’ve been here since the beginning or just found Swiss Army Mum, I’m glad you’re here.
Building sustainable health without overwhelm takes a village. If something resonated with you, I’d be grateful if you forwarded this to someone who might benefit or hit the ♥️ or ↻ Restack button. It helps more people discover this space.
Your Turn
Have you tried a CGM? What surprised you?
Which of the 10 hacks are you trying first?
Are you seeing patterns in your own glucose responses (even without a device)?
Comment below—I read every one.
See you next week.
— Mica
Lustig RH. Fructose: it's "alcohol without the buzz". Adv Nutr. 2013 Mar 1;4(2):226-35. doi: 10.3945/an.112.002998. https://pmc.ncbi.nlm.nih.gov/articles/PMC3649103/
de Andrade Mesquita L, Pavan Antoniolli L, Cittolin-Santos GF, Gerchman F. Distinct metabolic profile according to the shape of the oral glucose tolerance test curve is related to whole glucose excursion: a cross-sectional study. BMC Endocr Disord. 2018 Aug 16;18(1):56. doi: 10.1186/s12902-018-0286-7. https://pmc.ncbi.nlm.nih.gov/articles/PMC6097323/
Kim JY, Michaliszyn SF, Nasr A, Lee S, Tfayli H, Hannon T, Hughan KS, Bacha F, Arslanian S. The Shape of the Glucose Response Curve During an Oral Glucose Tolerance Test Heralds Biomarkers of Type 2 Diabetes Risk in Obese Youth. Diabetes Care. 2016 Aug;39(8):1431-9. doi: 10.2337/dc16-0352. https://pmc.ncbi.nlm.nih.gov/articles/PMC4955931/




