The Gut-Brain Connection: How Stress and Digestion are Locked in a Two-Way Conversation
What your gut is doing to your mood, and what your nervous system is doing back
We found each other on Substack the way most good things happen, by accident, and at the perfect time.
Erika writes The Regulated Mom. She’s a holistic health coach working with moms over 40 who struggle with anxiety, overstimulation and nervous system chaos. Her lane is the nervous system - specifically, what happens when it’s been stuck in overdrive for so long that chaos starts to feel like the norm. Dr. Mica writes Swiss Army Mum. She’s a scientist cutting through the noise for women 35+ who want the biology - the 20% of inputs that actually drive 80% of the outcomes - without the 12-step protocol or the overwhelm of a supplement stack.
We were writing about the same woman but from opposite ends of the same problem. Erika working top-down: regulate the nervous system, let the body follow. Mica working bottom-up: fix the biology, give the nervous system something solid to stand on.
The more we talked, the more we could see. These approaches don’t compete. They complete each other. They are two halves to the whole.
So we decided to write this together.
Today’s topic is the gut-brain axis. If you’ve ever noticed how a stressful week can trash your digestion, or noticed that eating like garbage makes your anxiety worse, you already know this connection exists. You just haven’t had anyone explain it from both ends at once.
Until now. Erika goes first.
Nervous System Dysregulation
Your Body Knew Before You Did. What Nervous System Dysregulation Looks Like in Midlife Moms
That’s the part nobody tells you. The anxiety that shows up out of nowhere. The bloating that comes and goes with zero logical pattern. The fact that you went from someone who could handle ANYTHING to someone who is one spilled cup away from a complete meltdown - and your stomach is somehow always involved.
That is not a coincidence. That is your nervous system talking to your gut, and your gut talking right back.
Here’s what’s happening.
When Your Nervous System is Running The Show, Your Gut Pays The Price
Chronic stress, the kind that doesn’t come from a single incident but from years of holding everything together for everyone, does something very specific to your digestive system. It is not subtle.
When your body perceives a threat, it shifts resources. Blood flow moves away from your digestive organs toward your muscles and heart. Gut motility slows, food sits, ferments, and becomes a problem. The migrating motor complex (MMC), responsible for keeping things moving and clearing bacteria between meals, gets disrupted. Research in The Journal of Physiology confirms that both acute and chronic stress produce wide-ranging changes across gut function - altered motility, increased sensitivity, disrupted intestinal signaling.1 This is where the bloating, constipation, nausea, and the “I don’t even know what’s happening in there” come from.
It gets messier. Chronic stress increases intestinal permeability. The tight junctions lining your gut loosen, and what’s supposed to stay contained - bacteria, toxins, partially digested food - starts leaking through. A 2023 study found that chronic stress significantly suppressed tight junction protein expression, increased gut permeability, and drove systemic inflammation as a result.2 Leaky gut is not just a wellness influencer trend. It is a documented physiological response to sustained stress.
And then there’s the microbiome. Mica will dive into how estrogen and the estrobolome factor in here as well. Stress reduces bacterial diversity, depletes beneficial strains, and creates conditions where harmful bacteria thrive. A 2023 analysis in Biological Psychiatry found that stress-driven microbiota shifts directly compromise the intestinal barrier, allowing bacterial products to enter the bloodstream and trigger systemic inflammation.3 Less microbial diversity means less production of short-chain fatty acids, or SCFAs, your gut lining needs to repair itself, fewer neurotransmitter precursors, and a more reactive, inflamed system overall.
What’s fascinating is your nervous system didn’t just stress you out. It physically restructured your gut.
You’ve been living the signs. You just didn’t know what you were reading.
The behavioral and emotional signs that the gut-brain axis is under strain are easy to miss because they don’t necessarily look like gut problems on the surface.
You’re anxious but can’t point to a reason.
Your mood crashes after meals.
Brain fog rolls in mid-afternoon and doesn’t lift.
You’re exhausted in a way that sleep doesn’t fix.
Food that never bothered you before is suddenly a problem.
You feel wired and depleted at the same time, which makes no sense and yet is somehow exactly how you feel.
Every. Single. Day.
There’s also the reactivity. The short fuse. The way your patience evaporates faster than it used to. That’s not a personality shift. That is a gut producing less serotonin because over 90% of your serotonin is made in the gut, produced by specialized cells in your intestinal lining, and regulated in part by your gut microbiome.4 And add in a nervous system that has been in threat mode so long it has forgotten what settled feels like.
This is what dysregulation looks like when it’s been going on long enough to become the baseline. You don’t feel stressed. You just feel like this is who you are now.
But it’s not.
Calming your nervous system is a gut intervention. Full stop.
The reason breathwork, rest, and co-regulation practices are not just stress management tools is because they directly shift the state of your gut. Vagal nerve activation, the kind that happens when your nervous system moves out of threat mode and into safety, restores gut motility, reduces intestinal permeability, and signals the microbiome environment that it is safe to function normally again. A 2022 study found that transcutaneous vagal nerve stimulation directly protected against stress-induced intestinal barrier breakdown in healthy adults.5
You cannot supplement your way out of a gut that is being dismantled by a nervous system stuck in survival mode. The regulation has to come first.
Now let’s flip the direction of the conversation. Erika showed what stress does to the gut.
Now Mica will show us what the gut does back to the brain.
Interested in The Regulated Mom? Start here:
Erika Glover is focused on helping moms over 40 with young kids go from anxiety, overstimulation, and mom rage to systemic regulation and finding safety and fun in motherhood again.
Here, we regulate first.
Your Gut Has 500 Million Neurons. Here Is What They Are Saying.
Here is the piece of the picture that changes how you read everything Erika just described: 90 to 95% of your serotonin is not made in your brain. It is made in your gut, by cells lining the intestinal wall, regulated by the bacteria living just beneath them.6 The neurotransmitter your mood, stress response, and cognitive clarity depend on originates, in the main, from below.
This is not a metaphor. It is anatomy. And it explains why regulating the nervous system from the top down, while your gut biology is quietly disrupted, is like trying to stabilize a building from the roof.
The wiring
The gut has its own local nervous system: 500 million neurons embedded in the gut wall that manage digestion completely independently, without waiting on instructions from the brain.7 This is the enteric nervous system, sometimes called the second brain. The vagus nerve is the long-distance cable that connects it to the brain. Think of it as a motorway with a very specific traffic pattern: roughly 80 to 90% of the vehicles are travelling upward, gut to brain, not the other way.8 Your gut is not a passive recipient of orders from headquarters. It is filing reports constantly, and most of the communication in this relationship originates below.
This is where the biology meets what Erika described. Chronic stress lands in the gut and disrupts it structurally. But the gut does not stay silent in response. Through the vagus nerve, through serotonin signalling, through the immune molecules produced by a disrupted microbiome, it sends that disruption straight back to the brain. The loop is genuinely bidirectional. Fixing one end without addressing the other is incomplete.
What perimenopause does to this loop
Oestrogen receptors are distributed throughout the gut: in the gut lining, in the enteric nervous system, and in the cells regulating the intestinal barrier. As oestrogen declines during perimenopause, the downstream effects reach well beyond hot flushes. A large study of 2,300 hispanic participants found that postmenopausal women had lower microbiome diversity and a significantly altered gut composition compared to premenopausal women: changes linked to worse cardiometabolic health.9
The bacteria responsible for metabolising and recirculating oestrogen, collectively known as the estrobolome, become less efficient as diversity drops, which drives oestrogen lower still. The loop tightens in the wrong direction. As the gut lining becomes more permeable, bacterial fragments cross into the bloodstream, reach the brain, and raise the baseline reactivity of the nervous system. It is one of the reasons mood strategies that worked reliably at 35 stop working at 45. The biological substrate has changed.
The metabolic amplifier
Chronically unstable blood sugar dysregulates the HPA axis (the body’s central stress-response system) over time, keeping cortisol elevated as the body repeatedly attempts to restore glucose homeostasis. Cortisol in turn directly disrupts the gut environment: it alters bacterial growth patterns, worsens gut permeability, and feeds the inflammatory loop the gut is already struggling to contain.10
Chronically elevated insulin deepens that inflammatory environment further. It builds silently over years, and one of the places it quietly does damage is the gut, where a chronically pro-inflammatory environment shifts the balance toward bacterial strains that worsen permeability and reduce short-chain fatty acid production.
Poor sleep is the third variable, and arguably the most underestimated. The gut operates on a tight circadian schedule. Epithelial cell proliferation, tight junction integrity, and microbial composition all oscillate in 24-hour cycles governed by the body’s internal clock.11 Sleep is the anchor of that clock. When it is fragmented or cut short, the circadian regulation of gut repair breaks down: barrier permeability increases, the timing of cellular renewal is disrupted, and the microbiome shifts toward dysbiosis
New here? Start here:
Swiss Army Mum is a practical guide to long-term health for busy women, built on four pillars: Body, Mind, Glow, and Flow.
Not every tool. Just the right ones.
The 80/20
The research converges on a short list of inputs that actually move the needle, and they map directly onto the variables above.
Fibre is the foundation. Most women are consuming roughly half the recommended 25 to 30 grams per day, which means the bacteria that ferment fibre into the short-chain fatty acids your gut lining depends on are working with a chronically depleted fuel supply.12 Vegetables, legumes, whole fruit, and whole grains are the sources. There is no supplement shortcut that replicates what a varied, fibre-rich diet does to microbial diversity over time.
Fermented foods add the diversity that perimenopause specifically erodes. A 2021 randomised controlled trial published in Cell, comparing a high-fibre diet to a high-fermented-food diet over ten weeks in human participants, found that fermented foods produced significantly greater increases in microbiome diversity.13 Variety of sources matters more than volume of any one: kefir, kimchi, sauerkraut, kombucha, miso. Rotate them.
Stable blood sugar protects the microbiome from cortisol-driven disruption. Protein at every meal, not skipping breakfast: these are glucose levers that are also gut levers.
And sleep is the repair window, not a lifestyle preference. The gut lining cannot sustain itself without it. That is physiology, not self-care advice.
The bottom line
A 2026 review set out to map what happens when declining oestrogen, gut microbiota dysbiosis, and anxiety converge in the same woman at the same time.14 What it found is what we have been describing from opposite ends of the same problem: these are not separate issues requiring separate fixes. They are one system, and the evidence now treats them that way.
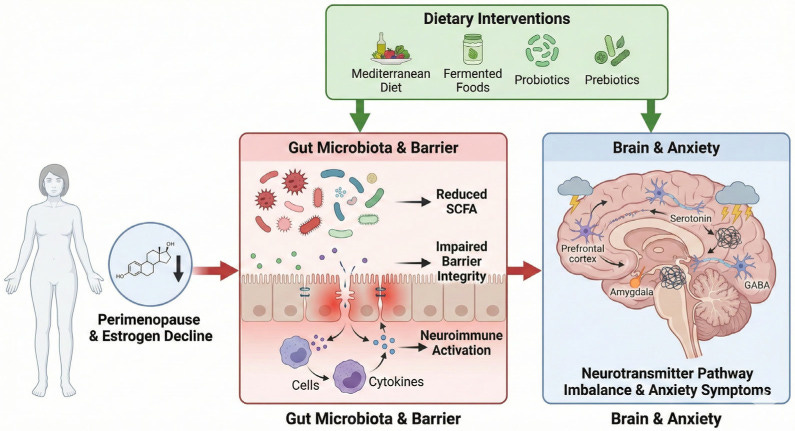
Neither is sufficient alone. Chronic dysregulation from the top keeps driving the gut disruption. Chronic gut inflammation from the bottom keeps undermining the nervous system work.
This is why so many women feel like they are doing “everything right” and still not feeling better. The nervous system and the gut are not separate projects. They are one conversation.
And in perimenopause, both sides get louder. The good news is that small interventions compound quickly when they target both ends of the loop simultaneously.
You do not need a complete overhaul.
You need enough safety for the nervous system to stand down, and enough biological support for the gut to stop sending distress signals back upstream.
That is where regulation starts feeling possible again.
Four things to start this week
You don’t need an overhaul. You need two things from each end of the axis, something that calms the system driving the disruption, and something that starts rebuilding what the disruption damaged. That’s it. Start integrating these 4 simple habits into your day this week, and see how it changes the way you feel.
One Screen-Free Meal a Day
Pick one meal, any meal, and eat it without an electronic - phone, TV, Kindle, etc. Just the food. Even if it’s just ten minutes.
This one sounds simple, but it is not easy. Most of us will gravitate toward grabbing our phones to scroll or check messages. But here’s what’s actually happening when you eat distracted: your nervous system stays in low-grade sympathetic activation, your digestive enzymes are suppressed, and your migrating motor complex - the mechanism responsible for clearing bacteria and moving food through your gut - never gets the parasympathetic signal it needs to function properly. You are eating while your gut is not fully engaged. Do that three times a day for years and the compounding effect is exactly what you’ve been feeling. One undistracted meal a day starts to reverse that. Nothing to do with the food, but because of the state you’re in when you eat it.
The 4-7-8 Breath Before You Eat
Before your next meal, before you pick up your fork, take 30 seconds to do this. Inhale for 4 counts. Hold for 7. Exhale slowly for 8. Do it twice.
That’s it.
Here’s why it matters. Your body cannot digest food properly in fight-or-flight. It was never designed to. When your nervous system is in threat mode, digestion is a low priority. Blood flow has already been rerouted, motility has slowed, and your gut is essentially on hold until it gets the signal that the threat has passed. The 4-7-8 breath sends that signal. It activates your vagus nerve, shifts you into parasympathetic state, and tells your gut it’s safe to do its job.
Fermented foods
Add one fermented food you actually like to one meal per day. Kefir in the morning, sauerkraut with lunch, kombucha with dinner. The goal is not quantity. It is consistency and variety over time. You are rebuilding diversity that perimenopause has been quietly eroding.
Savory breakfasts
Put protein at breakfast, every day this week, before anything else. This is the single fastest lever for blood sugar stability, cortisol regulation, and by extension, microbiome protection. It is also the meal most women skip or underserve. Eggs, Greek yoghurt, smoked salmon.
These four things address the system from both ends simultaneously. The nervous system calms. The gut gets substrate to work with. The inflammation has less fuel. The serotonin precursors have a better environment to do their job. This is what working together actually looks like, not two approaches running in parallel, but one system being supported from two directions at once.
References
Leigh, S.J., Uhlig, F., Wilkinson, L., et al. (2023). https://physoc.onlinelibrary.wiley.com/doi/full/10.1113/JP281951
Cheng, L., et al. (2023). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10454097/
Simpson, C.A., et al. (2023). https://www.sciencedirect.com/science/article/abs/pii/S0006322323016591
Yano, J.M., et al. (2015). https://doi.org/10.1016/j.cell.2015.02.047
Mogilevski, T., et al. (2022). https://pmc.ncbi.nlm.nih.gov/articles/PMC9787579/
Mawe GM, Hoffman JM (2013). https://doi.org/10.1038/nrgastro.2013.105
Furness JB. (2012) https://doi.org/10.1038/nrgastro.2012.32
Bonaz B, Bazin T, Pellissier S. (2018) https://doi.org/10.3389/fnins.2018.00049
Peters BA, Lin J, Qi Q, Usyk M et al. (2022) https://doi.org/10.1128/msystems.00273-22
Warren A, Nyavor Y, Beguelin A, Frame LA (2024) https://doi.org/10.3389/fimmu.2024.1365871
Voigt RM, Forsyth CB, Keshavarzian A (2019) https://doi.org/10.1080/17474124.2019.1595588
Dahl WJ, Stewart ML (2015). https://doi.org/10.1016/j.jand.2015.09.003
Wastyk HC, Fragiadakis GK, Perelman D et al. (2021) https://doi.org/10.1016/j.cell.2021.06.019
Marano G, d'Abate C, Ianes I et al. (2026) https://doi.org/10.3390/nu18050743







